
The next subshell to be filled is the 3 s subshell. Figure 8.15 “The 2 p Subshell.” For B through Ne, the 2 p subshell is being occupied. On the right side of the periodic table, these six elements (B through Ne) are grouped together (Figure 8.15 “The 2 p Subshell”). Figure 8.14 “The 2 s Subshell.” In Li and Be, the 2 s subshell is being filled.įor the next six elements, the 2 p subshell is being occupied with electrons. Figure 8.14 “The 2 s Subshell” shows that these two elements are adjacent on the periodic table. The next two electrons, for Li and Be, would go into the 2 s subshell. 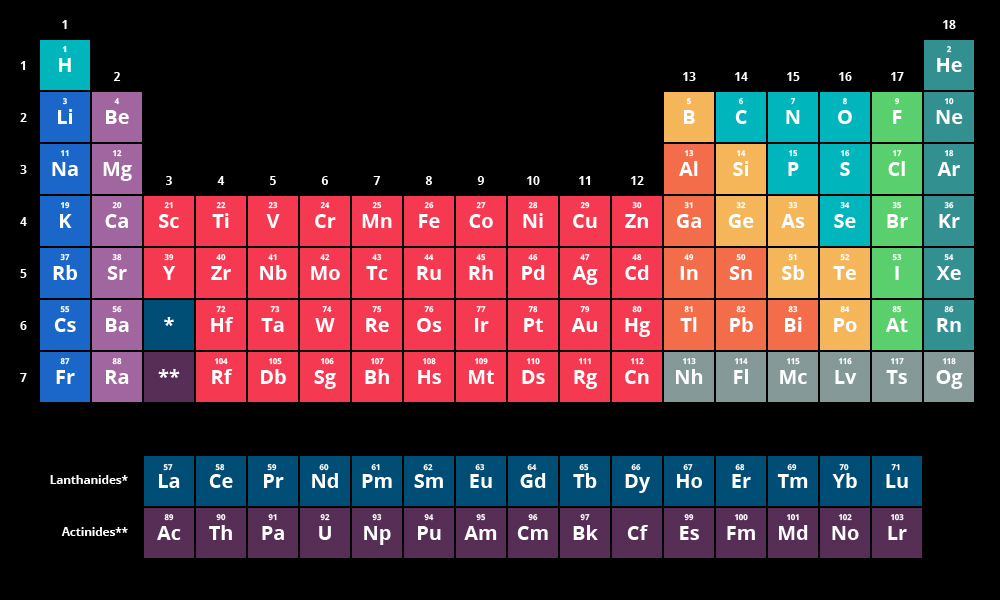
Figure 8.13 “The 1 s Subshell.” H and He represent the filling of the 1 s subshell. These two elements make up the first row of the periodic table (see Figure 8.13 “The 1 s Subshell”). Their electron configurations are 1 s 1 and 1 s 2, respectively with He, the n = 1 shell is filled. Why does the periodic table have the structure it does? The answer is rather simple, if you understand electron configurations: the shape of the periodic table mimics the filling of the subshells with electrons. Figure 8.12 “The Periodic Table.” View an accessible periodic table online. A periodic table is shown in Figure 8.12 “The Periodic Table.” The elements are listed by atomic number (the number of protons in the nucleus), and elements with similar chemical properties are grouped together in columns. In Chapter 3 “Atoms, Molecules, and Ions”, we introduced the periodic table as a tool for organizing the known chemical elements. Determine the expected electron configuration of an element by its place on the periodic table. 
Relate the electron configurations of the elements to the shape of the periodic table.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
