
Group 7 elements in the Periodic Table are known as the halogens, including fluorine (F), chlorine (Cl), bromine (Br), iodine (I), and astatine (At). These elements play crucial roles in various aspects of life, from supporting plant growth to being key components of semiconductor materials. Group 15 elements are called pnictogens, comprising nitrogen (N), phosphorus (P), arsenic (As), antimony (Sb), and bismuth (Bi). These elements share similar properties and find extensive applications in industries such as aerospace, automotive, and medical. The elements in Group 4, known as the titanium group, include titanium (Ti), zirconium (Zr), and hafnium (Hf). Group 4 Elements Are Known as Titanium Group Valence electrons are involved in chemical bonding and determine an element’s reactivity and ability to form compounds. The group number in the Periodic Table corresponds to the number of valence electrons an element possesses. Read also: 8 Astounding Facts About Carboxylic Acid The Group Number Indicates the Number of Valence Electrons These elements exhibit a wide range of properties and are used in various applications, from semiconductors to medicine. Group 13, also known as the boron group, includes elements such as boron (B), aluminum (Al), gallium (Ga), indium (In), and thallium (Tl). 
They often share similar chemical properties within their respective series. These elements have unique properties and are commonly referred to as the “rare earth” elements. The lanthanides and actinides are placed separately at the bottom of the Periodic Table. Lanthanides and Actinides – Separate Sections These metals are less reactive than the alkali metals but still have the tendency to form positive ions. Group 2 consists of the alkaline earth metals, such as beryllium (Be), magnesium (Mg), calcium (Ca), strontium (Sr), barium (Ba), and radium (Ra). Transition metals are known for their variable oxidation states and colorful compounds. They include elements such as iron (Fe), copper (Cu), zinc (Zn), silver (Ag), and gold (Au). The transition metals constitute a large portion of the periodic table and occupy multiple groups. They are highly reactive nonmetals and readily form compounds with various elements, especially alkali metals. 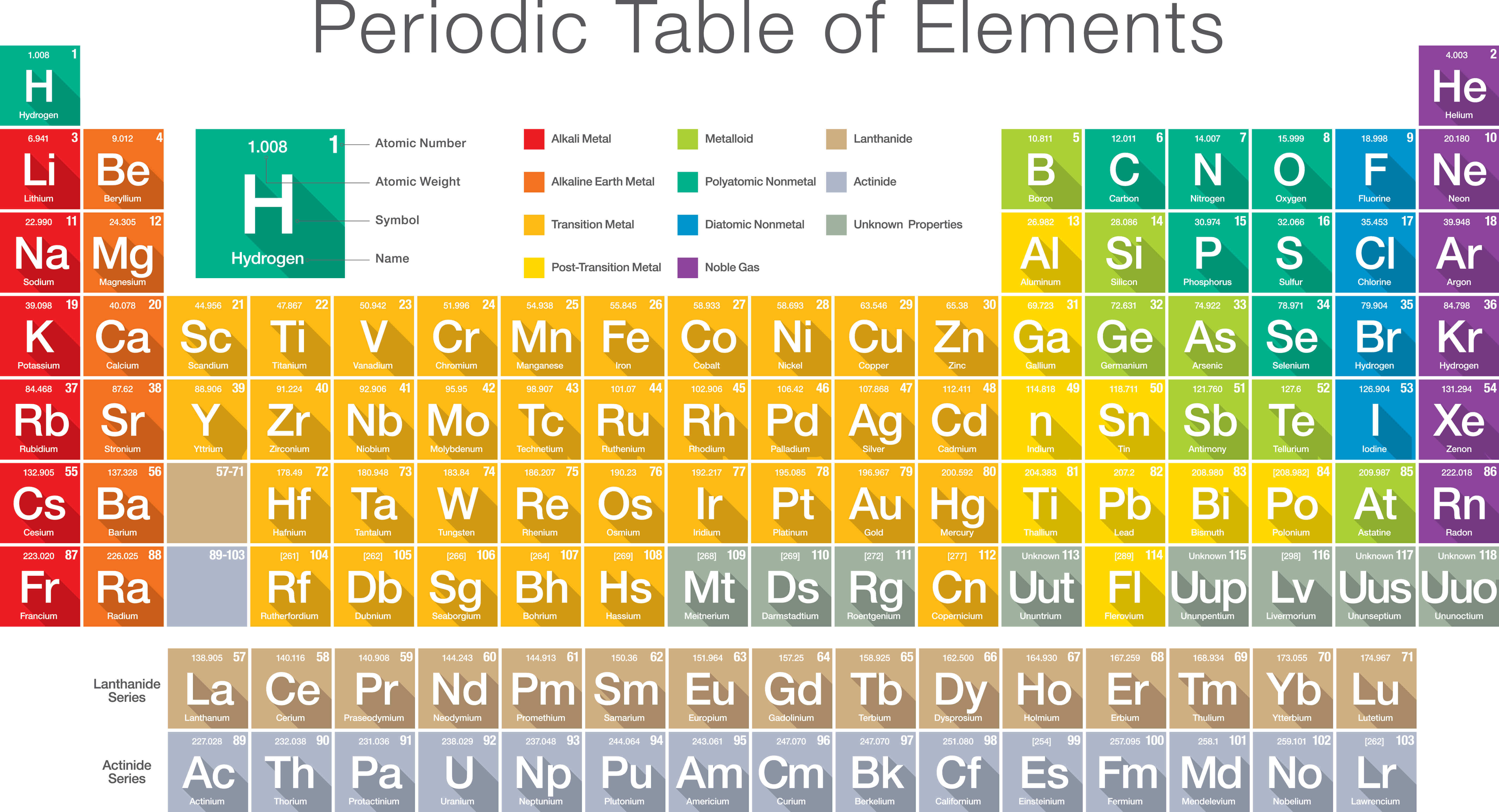
The elements in Group 17 are known as halogens, which include fluorine (F), chlorine ( Cl), bromine (Br), iodine (I), and astatine (At). Read also: 13 Fascinating Facts About Neutron Star Mergers Group 17 Elements Are Halogens They are highly reactive and can easily lose their outermost electron to form positive ions. Group 1 is occupied by the alkali metals, including lithium (Li), sodium (Na), potassium (K), rubidium (Rb), cesium (Cs), and francium (Fr). They are characterized by their low reactivity and full outer electron shells, making them stable and nonreactive. The elements in Group 18, also known as the noble gases, are helium (He), neon (Ne), argon (Ar), krypton (Kr), xenon (Xe), and radon (Rn). /GettyImages-1154261034-08fa91cb3d8942c093b9e6b66a26f690.jpg)
There are 18 groups in the modern periodic table, each designated by a number and a unique name. Group, also known as a family, is a vertical column in the Periodic Table that contains elements with similar properties and trends.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
